- patients
July 04, 2019
TFOS DEWS II Patient Summary
Dry eye disease affects millions of people throughout the world and is one of the most common reasons for patients to seek eye care. This symptomatic disease is characterized by an unstable and a more concentrated (termed
hyperosmolar) tear film, which leads to increased inflammation and damage to eye structures and nerves. Moderate to severe dry eye can be life-altering and may be associated with eye pain, limitations in performing daily activities (e.g. reading, driving, use of digital devices), sleep deprivation, reduced energy, poor general health, and often depression.
Two forms of dry eye are recognized, aqueous deficient resulting from reduced tear secretion, and evaporative resulting from excessive tear evaporation. The core mechanism of dry eye disease is evaporation-induced tear
hyperosmolarity (higher salt concentration than normal), which is the trigger for a cascade of events leading to eye surface damage and inflammation. The most common cause of evaporative dry eye is meibomian gland dysfunction.
A recommended sequence of tests for the diagnosis of dry eye disease first utilizes questions to exclude conditions that mimic dry eye. A dry eye diagnosis then requires a positive score on one of two specific symptom questionnaires, followed by at least one positive clinical sign indicating a reduced non-invasive tear break-up time (a measure of tear film stability), an elevated or a large inter-eye disparity in osmolarity (tear saltiness), or ocular surface damage indicated by dye staining.
The management of dry eye disease can be challenging due to its multifactorial etiology. Determining the major causative factors behind the dry eye is critical to selecting the appropriate management. The ultimate goal of dry eye management is to restore the homeostasis of the surface of the eye and tear film. While certain treatments may be specifically indicated for one aspect of a patient’s condition, a number of therapies may be appropriate to manage a patient presenting with dry eye. The treatment of dry eye remains something of an art, requiring an individualized approach for affected patients. There is no single approach to dry eye management that will suit all patients.
Contents
1. Global effort to increase understanding of dry eye disease
2. Classification and definition of dry eye disease
3. Epidemiology of dry eye disease
4. Role of sex, gender, and hormones in dry eye disease
5. Pathophysiology of dry eye disease
6. Tear film in dry eye disease
7. Pain and sensation in dry eye disease
8. Iatrogenic dry eye disease
9. Diagnosis of dry eye disease
10. Management and therapy of dry eye disease
11. Design of clinical trials for dry eye disease treatment
12. Glossary of highlighted terms
13, Dedication
14. Acknowledgments
15. References |
Global Effort To Increase Understanding Of Dry Eye Disease1
To increase our understanding of dry eye disease, the Tear Film & Ocular Surface Society (TFOS) launched the Dry Eye Workshop II (TFOS DEWS II) in March 2015, which lasted for more than two years, and involved the efforts of over 150 eye doctors and researchers around the world. The reason that TFOS sponsored and organized this initiative is because TFOS is a non-profit organization with a mission to advance eye and tear film research, knowledge and education. The goal of TFOS DEWS II was to achieve consensus among the diverse members to 1) update the definition and classification of dry eye; 2) clarify the patterns, causes and effects of the disease; 3) provide recommendations for the diagnosis, management and treatment of dry eye, and 4) delineate clinical trial design for testing new therapies for dry eye. The entire TFOS DEWS II report is available on the TFOS website (http://
www.TearFilm.org) and the TFOS DEWS II App (http://www.tearfilm.org/dettnews-download_the_tfos_dews_ii_app__view_tfos_dews_ii_videos/5602_16/eng/). This article presents a patient-focused Executive Summary of the conclusions and recommendations of the TFOS DEWS II Subcommittee reports. A glossary of terms appears at the end of this article. The authors of this Summary are cited in the reference list.
2
Classification And Definition Of Dry Eye Disease3
Evidence supports a classification scheme that accounts for elements of both aqueous deficient and evaporative types of dry eye in the diagnosis and management of the disease. Aqueous deficient dry eye refers to lacrimal gland dysfunction, and evaporative dry eye includes both eyelid-related causes such as meibomian gland dysfunction (MGD), inadequate lid closure during sleep (
nocturnal lagophthalmos) and blink-related problems, and conditions related to the surface of the eye (e.g. changes to mucins or lipids).
Dry eye has both symptoms experienced by the patient and signs detected during clinic-based testing and can be differentiated from other eye diseases by a careful examination of tell-tale signs. Symptoms may include changes in vision as well as symptoms of discomfort, such as dryness, grittiness, and burning.
Figure 1 represents a clinical decision-making tree or flow chart, which begins with an assessment of patient symptoms, and is followed by the identification of signs of eye surface disease.
The lower portion of
Figure 1 highlights the two main dry eye categories: aqueous deficient and evaporative. Evidence suggests that dry eye is mostly evaporative in nature. While it is possible that either type can occur without obvious signs of the other, as the disease progresses, it is increasingly likely that characteristics of both types of dry eye will become evident.
The goal was to create an updated and
evidence-based definition and a contemporary classification of dry eye disease to help guide clinical management and future research. The new definition is
“Dry eye is a multifactorial disease of the ocular surface characterized by a loss of homeostasis of the tear film, and accompanied by ocular symptoms, in which tear film instability and hyperosmolarity, ocular surface inflammation and damage, and neurosensory abnormalities play etiological roles.”
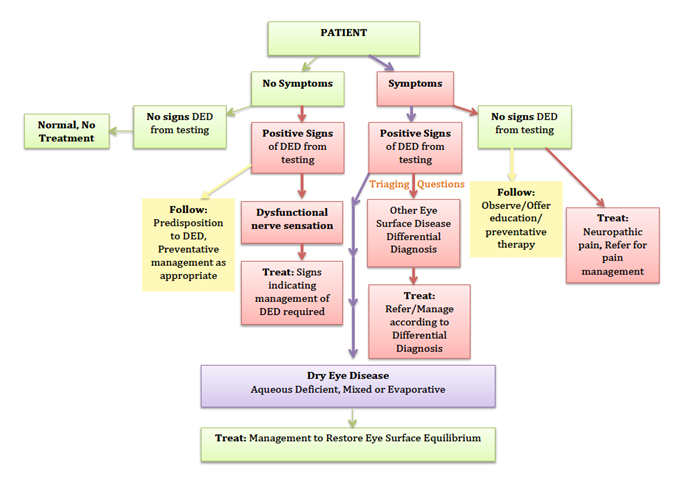 Figure 1.
Figure 1. Classification of dry eye disease
Homeostasis refers to balance, hyperosmolarity to the tear film being more concentrated (i.e. too salty), neurosensory abnormalities to change in the sensitivity of the ocular surface caused by the damage, with etiological roles indicating what causes dry eye.
Epidemiology Of Dry Eye Disease4
The overall prevalence of dry eye with and without symptoms ranges from 5% to 50%, while the prevalence based on signs of eye surface disease alone is generally higher and more variable, reaching up to 75% in some populations.
Risk factors for having dry eye are summarized in
Table 1.
| Table1: Dry Eye Disease Risk Factor Categorization |
| Consistent |
Probable |
Inconclusive |
| aging |
diabetes |
Hispanic ethnicity |
| female sex |
rosacea |
menopause |
| Asian race |
viral infection |
acne |
| meibomian gland dysfunction |
thyroid disease |
sarcoidosis |
| connective tissue disease |
psychiatric conditions |
smoking |
| Sjögren syndrome |
pterygium |
alcohol |
| androgen deficiency |
low fatty acid intake |
pregnancy |
| computer use |
refractive surgery |
demodex infestation |
| contact lens wear |
allergic conjunctivitis |
botulinum toxin injection |
| use of medications, such a, antihistamines, antidepressants, anxiolytics, and isotretinoin |
medications (for example, anti-cholinergics, diuretics, ? diuretics |
multivitamins |
| estrogen replacement therapy |
|
oral contraceptives |
| hematopoietic stem cell transplantation |
|
|
| environmental conditions, such as pollution, low humidity, and sick building syndrome |
|
|
The signs and symptoms of dry eye increase with age, however the presence of signs detected through eye testing show a greater increase per decade of life than symptoms. Few studies have been conducted in populations under the age of 40 where dry eye is also present, and some signs of dry eye may be related to normal aging. Higher rates of dry eye occur in women than men, although the differences generally only become significant with increasing age.
The influence of dry eye on the individual is considerable given its detrimental effect on vision, quality of life, work productivity, as well as the psychological and physical effects of pain. The financial burden of dry eye on the individual and society is substantial, the most significant impact being indirect costs due to reduced work productivity.
Future research needs to include better evaluation of the prevalence of dry eye of differing severity and in youth, the incidence in varied populations, the impact of modifiable risk factors such as mobile device usage, the influence of climate, environment and socioeconomic factors, and the natural history of both treated and untreated dry eye disease.
Role Of Sex, Gender, And Hormones In Dry Eye Disease5
One of the most compelling features of dry eye disease is that it occurs more frequently in women, such that being of female sex significantly increases the risk factor of dry eye.
Many reported sex-related differences are attributed to the effects of androgens (hormones), as they are extremely important in regulation of the eye surface and surrounding tissues; their androgen deficiency can lead to development of both types of dry eye. Sex-related differences may also be attributed to the genes on the sex chromosomes.
It is important to note that the word “sex” is used for a reason. “Sex” refers to the classification of living things, generally as male or female, according to their reproductive organs and functions assigned by chromosomes. “Gender,” on the other hand, refers to a person’s self-representation as a man or woman, or how social institutions respond to that person based on the individual’s gender presentation.
Both sex (female/male) and gender (feminine/masculine) affect dry eye risk, how dry eye presents, the immune system's response, perceived pain level, care-seeking behavior and interactions with eye care professionals.
Pathophysiology Of Dry Eye Disease6
The core mechanism of dry eye disease is evaporation-induced tear hyperosmolarity (higher salt concentration than normal), which is the trigger for a cascade of events leading to eye surface damage and inflammation.
Two forms of dry eye are recognized, aqueous deficient resulting from reduced tear secretion, and evaporative resulting from excessive tear evaporation due to a dysfunctional tear film (e.g. changes to lipids on the surface of the tear film).
Tear film instability can be initiated by conditions that affect the surface of the eye, such as vitamin A deficiency, eye allergies, use of preservatives in topical medications, contact lens wear, certain cosmetics, low humidity, blowing air and computer vision syndrome (see Tables 1 and 2).
.jpg) Figure 2.
Figure 2. Pathophysiology of dry eye disease
Aqueous deficient dry eye may result from blocking the sensory drive to the lacrimal gland, chronic topical anesthetic use, reduced reflex tearing due to nerve damage or refractive surgery (such as LASIK surgery), obstruction to the lacrimal ducts, a number of systemic drugs such as antihistamines, b-blockers, bladder and bowel
antispasmodic agents, diuretics, and specific psychotropic drugs, or aqueous tear reduction due to aging.
In the Western world the most common cause of aqueous deficient dry eye is inflammation of the lacrimal gland, as seen in autoimmune disorders such as Sjögren syndrome.
The most common cause of evaporative dry eye is MGD, which results in low delivery of lipid to the surface of the eye. The most common cause of MGD, in turn, is obstruction of the meibomian gland's external duct, which leads to tear film instability, increased tear evaporation and ultimately to evaporative dry eye.
The prevalence of MGD increases after the age of 50 years, a process that may be linked to a decrease in
bioavailable androgens (hormones). Use of cis-retinoic acid (for example, isotretinoin or retinoic acid treatment of acne vulgaris) and certain anti-glaucoma eye drops may induce MGD. A variety of disorders, such as acne rosacea and psoriasis, are associated with MGD.
Both evaporative and aqueous deficient dry eye can lead to friction-related symptoms and ocular surface damage.
Tear Film In Dry Eye Disease7
Evidence supports a two-layered model of the tear film, involving a thin surface lipid layer overlying a thicker mucous-aqueous mixed layer. The mucin component of this latter layer helps to wet the eye’s surface, which in turn allows the watery aqueous component to spread over an otherwise non-wetting surface.
While the extent of the role of the tear film lipid layer alone in preventing evaporation and breakup of tears is unclear, it is likely that interactions of the whole tear film, including lipids, mucins, proteins and salts, prevent evaporation and collapse of the tear film. Several studies have attempted to correlate changes in tear lipid biochemistry with dry eye, but no definitive links
Tear hyperosmolarity is the hallmark of dry eye disease, Tear film osmolarity increases with dry eye severity.
A holistic approach to understanding tear film structure and function, together with improvements in characterizing tear film biochemistry, are expected to lead to identification of new markers that can be used to diagnose, potentially predict, and even treat dry eye.
Pain And Sensation In Dry Eye Disease8
Pain is differentiated into two types. The first type is
nociceptive, which is pain in response to actual damage to tissues. Pain when you burn your hand on the stovetop is
nociceptive pain. The second type of pain is
neuropathic, which is pain due to an abnormality anywhere along the pathway of nervous system that conducts sensation. Phantom limb pain is an example
neuropathic pain.
Pain associated with dry eye is transmitted via nerve pathways from the ocular surface to the brain.
Tear evaporation between blinks causes distinct cooling of the eye and increases the osmolarity of tears. This increased osmolarity triggers the activity of cold-sensing nerve receptors in the cornea and contributes to the reflex control of tear production and eye blinking. Specific regions in the brain stem play a dominant role in sensing the osmolarity of tears and ocular pain, which encourages maintenance of tear film stability, which in turn helps to alleviate eye pain.
Reduced tear secretion in dry eye leaves the cornea exposed to adverse environmental conditions, which can lead to varying levels of inflammation and to nerve damage.
Iatrogenic Dry Eye Disease9
Dry eye disease can be caused by a variety of medical interventions (termed
iatrogenic), including the use of topical and systemic drugs, preservatives, contact lenses, and exposure to ophthalmic surgical and non-surgical procedures.
| Table 2: Topical And Systemic Medications That May Induce Or Worsen Dry Eye Disease |
| Topical Medication Categories |
Systemic Medication Categories and Sub-Categories |
| Adrenergic agonists |
Analgesics: Antirheumatic, Cannabinoid, Opioid |
| Antiallergics |
Anticholinergic (antimuscarinics): Antiarrythmic/Bronchodilating, Antihistamine, Antidepressant, AntiParkinson’s, Antipsychotic, Antispasmodic, Decongestant |
| Antivirals |
Antihypertensives: Adrenergic blocking
Na+Cl- Co-transporter (diuretic) |
| b?Adrenergic receptor blockers |
Hormonal: Antiandrogen/Estrogen replacement |
| Carbonic anhydrase inhibitors |
Anesthesia |
| Cholinergic agonists |
Antileprosy |
| Decongestants |
Antimalarial |
| Miotics |
Antineoplastic |
| Mydriatics & cyclopegics |
Anxiolytic/hypnotic |
| Prostaglandins |
Chelator/Calcium Regulator |
| Topical and local anesthetics |
Depressant |
| Topical ocular non-steroidal anti-inflammatory drugs |
Herbal and Vitamins |
| |
Neurotoxin |
| |
Sedative |
Contact lens wear has been identified as causing or being associated with dry eye. Changes to the tear film in contact lens wearers with dry eye include lipid layer thinning, tear film instability, lower tear turnover and a decreased volume of tears on the ocular surface.
Corneal refractive surgery, corneal transplantation, cataract surgery, eyelid surgery, cosmetic procedures and botulinum toxin application may cause or aggravate dry eye.
Topical and systemic medications and drug groups that may induce or worsen dry eye are listed in
Table 2.
More research is needed to identify dry eye risk factors, detect early dry eye prior to eye surgery, determine the benefits of proactive pretreatment, and to develop less toxic medications and less disruptive eye surgeries.
Diagnosis of Dry Eye Disease10
A recommended sequence of tests for the diagnosis of dry eye disease and an assessment of its severity and the relative contributions of the aqueous deficient or evaporative dry eye subtypes are shown in Figure 3 below.
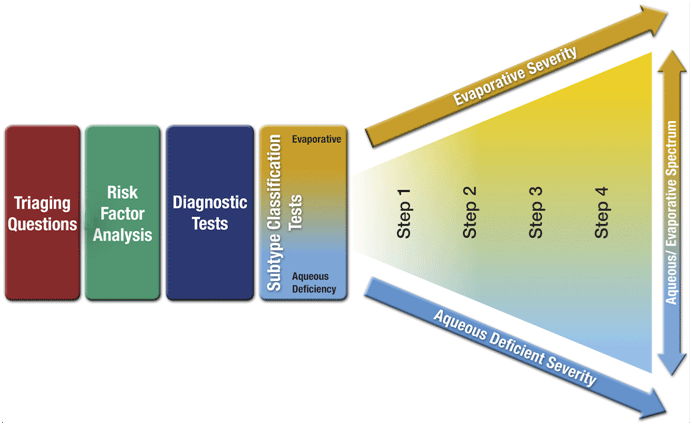 Figure 3.
Figure 3. Dry eye disease diagnosis and management
This diagnostic process first utilizes questions to exclude conditions that mimic dry eye. A dry eye diagnosis then requires a positive score on one of two specific symptom questionnaires, followed by at least one positive clinical sign indicating a reduced non-invasive tear break-up time (a measure of tear film stability), an elevated or a large inter-eye disparity in osmolarity (tear saltiness), or ocular surface damage indicated by dye staining.
After confirming that the condition is dry eye, further subtype classification tests such as imaging the meibomian glands, observing the lipid layer on the surface of the tears, and tear volume measurement should be performed to determine: [a] where the dry eye falls on the spectrum between aqueous deficient and evaporative, and [b] the severity of dry eye, as these help to guide treatment (see below).
Management and Therapy of Dry Eye Disease11
The management of dry eye disease can be challenging due to its multifactorial
etiology. Determining the major causative factors behind the dry eye is critical to selecting the appropriate management.
The ultimate goal of dry eye management is to restore the homeostasis of the surface of the eye and tear film. While certain treatments may be specifically indicated for one aspect of a patient’s condition, a number of therapies may be appropriate to manage a patient presenting with dry eye.
Although a priority is to identify and manage the primary source of the disease, the management of dry eye invariably involves prolonged therapy to address chronic signs and symptoms.
Scientific evidence, as well as risk/benefit and cost considerations, will also contribute to decisions made in choosing between multiple treatment options.
While there is strong evidence that individual treatments are more effective at managing dry eye than no treatment, there is little information to suggest the dry eye subtype and severity for which they are most beneficial. This is therefore the focus of ongoing research.
The evidence supports a staged management and treatment of dry eye (Table 3).
| Table 3: Recommendations for the staged management of dry eye disease |
Step 1:
- Education regarding the condition, its management and prognosis
- Modification of local environment (e.g. humidity, pollution)
- Education regarding potential dietary modifications (including oral essential fatty acid supplementation)
- Identification and potential modification/elimination of offending systemic and topical medications
- Ocular lubricants of various types (if MGD is present, then consider lipid-containing supplements)
- Lid hygiene and warm compresses of various types
|
Step 2:
If above options are inadequate consider:
- Non-preserved ocular lubricants to minimize preservative-induced toxicity
- Tea tree oil treatment for Demodex (if present)
- Tear conservation
- Punctal occlusion
- Moisture chamber spectacles/goggles
- Overnight treatments (such as ointment, moisture chamber devices or gentle tape holding eyelids shut while sleeping)
- In-office, physical heating and expression of the meibomian glands (including device-assisted therapies)
- In-office intense pulsed light therapy for MGD
- Prescription drugs to manage dry eye disease
- Topical antibiotic or antibiotic/steroid combination applied to the lid margins for anterior blepharitis (if present)
- Topical corticosteroid (limited-duration)
- Topical secretagogues
- Topical non-glucocorticoid immunomodulatory drugs (such as cyclosporine)
- Topical LFA-1 antagonist drugs (such as lifitegrast)
- Oral macrolide or tetracycline antibiotics
|
Step 3:
If above options are inadequate consider:
- Oral secretagogues
- Autologous/allogeneic serum eye drops
- Therapeutic contact lens options
- Soft bandage lenses
- Rigid scleral lenses
|
Step 4:
If above options are inadequate consider:
- Topical corticosteroid for longer duration
- Amniotic membrane grafts
- Other surgical approaches (e.g. punctal occlusion; tarsorrhaphy, salivary gland transplantation)
|
The anticipated therapy duration is related to the individual's compliance and response and to the treatment being considered. Most often, therapeutic effects are observed within one to three months, although some treatments (e.g. cyclosporine A) may take longer.
Overall, the treatment of dry eye remains something of an art, requiring an individualized approach for affected patients. There is no single approach to dry eye management that will suit all patients.
Design Of Clinical Trials For Dry Eye Disease Treatment12
In order to improve the quality of dry eye clinical trials, to optimize resources, and to improve patient access to novel treatments, the following recommendations are made:
- Studies should be conducted under Good Clinical Practice guidelines, which requires compliance with appropriate regulatory requirements.
- The Consolidated Standards of Reporting Trials (CONSORT; http://www.consort-statement.org/) statement is useful to review prior to planning and starting a study.
- The trial design and sample size should align with the type of treatment under investigation, the purpose of the study, and the stage of development.
- The dose of a therapy must be non-toxic, but be sufficiently strong and frequently administered to achieve the optimal therapeutic results.
- The duration of treatment, at least for a pivotal study (which is one that provides evidence for getting regulatory approval to commercialize a drug), should correspond with the drug’s mechanism of action and the time taken to achieve its desired effect.
- For pivotal studies, number of trial subjects is key to the potential validity of the study.
- Test choice is critical in confirming how well a treatment works., If possible, test procedures should be minimally invasive and non-operator dependent, to avoid biasing the results, as well as being relevant to the changes that are anticipated to occur with administration of the treatment.
- Exploration of novel ways to evaluate dry eye disease, such as biomarker evaluation, may lead to improvement in dry eye clinical trial design and increased clarity on the efficacy of new treatments.
Glossary Of Highlighted Terms
Allogeneic - of cells or tissues obtained from a genetically similar, but not identical, donor
Anterior blepharitis - inflammation around the eyelid skin, lashes, and lash follicles
Antispasmodic agents – medicines used to treat symptoms such as tummy pain and cramp (spasm). They are most commonly used for symptoms of irritable bowel syndrome
Autologous - of cells or tissues obtained from the same individual
Bioavailable - the proportion of a drug or other substance that enters the circulation when introduced into the body and so is able to have an active effect
Epidemiology – the branch of medicine relating the incidence, distribution, and possible control of diseases and other factors relating to health
Etiology - the cause of a disease
Evidence-based - any concept or strategy that is derived from or informed by unbiased scientific evidence
Homeostasis - the tendency of the body to seek and maintain a condition of balance within its internal environment
Hyperosmolar or Hyperosmolarity – referring to the increased osmolar concentration (saltiness) of body fluids
Iatrogenic - relating to illness caused by medical examination or treatment
Lipid Layer - a blanket of fats or oils that helps slow tear or water evaporation
Neuropathic - pain due to an abnormality anywhere along the pathway of nervous system that conducts sensation. Phantom limb pain is classified as a neuropathic pain.
Nociceptive - pain in response to actual damage to tissues. Pain when you burn your hand on the stovetop is nociceptive pain.
Nocturnal lagophthalmos - refers to the inability of apparently closed lids to exclude air from the ocular surface during sleep. It may be responsible for dry eye symptoms occurring immediately upon rising
Pathophysiology - functional changes that accompany a disease
Serum - protein-rich liquid that separates out when blood coagulate
Dedication
This TFOS DEWS II report is dedicated to the late Professor Juha Holopainen (Helsinki Eye Lab and Department of Ophthalmology, University of Helsinki and Helsinki University Hospital, Helsinki, Finland), who served on the TFOS DEWS II Steering Committee and Tear Film Subcommittee, in recognition of his outstanding scientific contributions to the field of the ocular surface and tear film.
Acknowledgments
The authors thank all participants of TFOS DEWS II, and the TFOS staff and consultants for their contributions to this report.
The TFOS DEWS II was supported by unrestricted donations from Alcon, Novartis, Shire, Allergan, Bausch+Lomb, Akorn, CooperVision, Dompé, Horus Pharma, Lµbris Biopharma, Oculeve, TearLab, Laboratoires Théa, SIFI, Sun Pharma, Johnson & Johnson VisionCare, Carl Zeiss Meditec, Quint Health, Scope Ophthalmics and Senju.
 Figure 6.
Figure 6. TFOS DEWS II
TM report
13
References
1. Nelson JD, Craig JP, Akpek E, Azar DT, Belmonte C, Bron AJ, et al. TFOS DEWS II Introduction. Ocul Surf 2017;15:269-75.
2. The authors of this article are: Nelson, J Daniel (Associate Medical Director for Specialty Care for HealthPartners Medical Group and Clinics, St Paul, MN, USA); Craig, Jennifer (Associate Professor, University of Auckland School of Medicine, Auckland, New Zealand); Jones, Lyndon (Director, Centre for Ocular Research & Education, and Professor of Optometry, University of Waterloo, Ontario, Canada); Stapleton, Fiona (Professor and Head, School of Optometry and Vision Science, University of New South Wales, Sydney, Australia): Willcox, Mark (Professor, School of Optometry and Vision Science, University of New South Wales, Sydney, Australia); Wolffsohn James (Professor and Associate Pro-Vice Chancellor, Aston University, Aston, UK); Sullivan, Amy Gallant (TFOS Executive Director, Boston, MA, USA); Levesque, Allison Porreco (TFOS Community Advocate, Carlsbad, CA, USA); Hammitt, Katherine (Vice President of Medical and Scientific Affairs, Sjögrens Syndrome Foundation, Reston, VA, USA); Bitton, Etty (Associate Professor, Director of Dry Eye Clinic, École d´Optométrie, Université de Montréal, Montreal, Quebec, Canada); Cohen, Stephen (Optometrist, Doctor My Eyes, Scottsdale, AZ, USA); Epstein, Arthur (Director, Clinical Research, and Head, Dry Eye - Ocular Surface Disease Center, Phoenix Eye Care, AZ, USA); Gupta, Preeya K (Assistant Professor of Ophthalmology, Duke University School of Medicine, Durham, NC, USA); Marini, Cecilia (Medical Staff & Coordinator, Corneal & Ocular Surface and Anterior Segment Services, and Chief, Corneal Transplant Team, El Cruce Hospital, Buenos Aires, Argentina); O'Dell, Leslie (Director of the Dry Eye Center of PA at Wheatlyn Eye Care, York, PA, USA); Parsloe, Colin (Consultant, Milton Keynes General NHS Trust, Luton, UK); Perry, Christina (Dry eye patient, Chapel Hill, NC, USA); Shen, Joanne (Assistant Professor and Chair, Department of Ophthalmology, and Director of the Dry Eye Clinic, Mayo Clinic, Scottsdale, AZ, USA; Suh, Leejee H (Associate Professor of Ophthalmology, Columbia University, and Director, Cornea and Refractive Surgery Division, Harkness Eye Institute, New York City, NY, USA); Starr, Christopher (Associate Professor of Ophthalmology, Weill Cornell Medical College, and Associate Attending Ophthalmologist, New York-Presbyterian Hospital, New York City, NY, USA); Sullivan, David A (Senior Scientist, Schepens Eye Research Institute & Massachusetts Eye and Ear, and Associate Professor, Harvard Medical School, Boston, MA, USA)
3. Craig JP, Nichols KK, Akpek EK, Caffery B, Dua HS, Joo CK, et al. TFOS DEWS II De?nition and Classi?cation report. Ocul Surf 2017;15:276-83.
4. Stapleton F, Alves M, Bunya VY, Jalbert I, Lekhanont K, Malet F, et al. TFOS DEWS II Epidemiology report. Ocul Surf 2017;15:334-65.
5. Sullivan DA, Rocha EM, Aragona P, Clayton JA, Ding J, Golebiowski B, et al. TFOS DEWS II Sex, Gender, and Hormones report. Ocul Surf 2017;15: 284-333.
6. Bron AJ, dePaiva CS, Chauhan SK, Bonini S, Gabison EE, Jain S, et al. TFOS DEWS II Pathophysiology report. Ocul Surf 2017;15:438-510.
7. Willcox MDP, Argüeso P, Georgiev G, Holopainen J, Laurie G, Millar T, et al. TFOS DEWS II Tear Film report. Ocul Surf 2017;15:366-403.
8. Belmonte C, Nichols JJ, Cox SM, Brock JA, Begley CG, Bereiter DA, et al. TFOS DEWS II Pain and Sensation report. Ocul Surf 2017;15:404-37.
9. Gomes JAP, Azar DT, Baudouin C, Efron N, Hirayama M, Horwath-Winter J, et al. TFOS DEWS II Iatrogenic Dry Eye report. Ocul Surf 2017;15:511-38.
10. Wolffsohn JS, Arita R, Chalmers R, Djalilian A, Dogru M, Dumbleton K, et al. TFOS DEWS II Diagnostic Methodology report. Ocul Surf 2017;15:539-74.
11. Jones L, Downie LE, Korb D, Benitez-del-Castillo JM, Dana R, Deng SX, et al. TFOS DEWS II Management and Therapy report. Ocul Surf 2017;15:575-628.
12. Novack GD, Asbell P, Barabino B, Bergamini MVW, Ciolino JB, Foulks GN, et al. TFOS DEWS II Clinical Trial Design report. Ocul Surf 2017;15:629-49.
13. Craig JP, Nelson JD, Belmonte C, Bron AJ, Chauhan S, de Paiva CS, Gomes J, Nichols K, Novack GD, Stapleton FJ, Willcox MDP, Wolffsohn JS, Sullivan DA. TFOS DEWS II Report Executive Summary. Ocul Surf 2017;15:802-812.



.jpg)











